Description
Synonyms (rus): Osmotic resistance of erythrocytes, spherocytosis, thalassemia, hemolytic anemia
Synonyms (eng): Osmotic fragility, spherocytosis, thalassemia, hemolytic anemia
Biomaterial: Venous blood
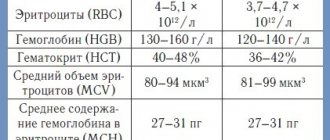
Indicator(s): Osmotic resistance of red blood cells
Method(s): Osmotic lysis
Container type and preanalytical features: Hematology test tube with EDTA, 2 ml (purple cap). The test for osmotic stability of red blood cells is performed in freshly prepared blood (within 2 hours after collection), as well as after incubation at 37 ° C for 24 hours.
The red blood cell osmotic stability test is used to measure the resistance of red blood cells to hemolysis when exposed to different concentrations of saline solution. When red blood cells are exposed to a hypotonic environment, water enters the cell and causes cell lysis. The red blood cell osmotic stability test is a classic test for diagnosing hereditary spherocytosis. Spherocytes are osmotically less stable cells that are more easily lysed in a hypotonic solution compared to normal erythrocytes. The classic osmotic resistance test, originally described by Parpart et al. (1947), consists of adding whole blood to a series of saline solutions ranging from 0.1% to 0.9%. These samples are centrifuged and absorbance measured at 540 nm to calculate the percentage of hemolysis for each solution.
Method for preparing leukofiltered washed erythrocytes
V.N. Melnikova, E.A. Selivanov, G.Yu. Kiryanova, T.A. Efimova
Russian Research Institute of Hematology and Transfusiology, St. Petersburg
Transfusiology No. 3, 2010
A method has been proposed for preparing an erythrocyte medium that is maximally free of impurities of leukocytes, platelets and plasma by leukodepletion using a domestic leukofilter device ULL-01 and subsequent washing with an isotonic sodium chloride solution, which makes it possible to increase its immunological and infectious safety. The possibility of storing this medium at 4°C in the erythropreservative SAGM has been substantiated.
Key words: erythrocytes, leukodepletion, laundering, morphofunctional properties, safety.
Introduction
The principle of blood component therapy involves the use of blood transfusion media, maximally purified from “ballast” elements, for the correction of various pathological conditions.
The purpose of transfusions of erythrocyte-containing media is to correct the anemic syndrome, therefore allogeneic leukocytes - carriers of highly immunogenic antigens (HLA system and specific), not required for this category of patients, can cause post-transfusion non-hemolytic temperature reactions, acute pulmonary failure, and cause alloimmunization , platelet refractoriness, immunosuppression, and also become a source of leukocyte-associated viral infections.
In addition, the admixture of leukocytes serves as a source of accumulation in erythrocyte media of cytokines and proteolytic enzymes, released from rapidly degrading white cells and having a membranotropic (hemolytic) effect, which significantly worsens the conditions of their storage. The presence of leukocytes and platelets in the erythrocyte environment serves as the basis for the formation of microclots.
Despite the fact that the most immunogenic blood fraction is leukocytes, the presence of plasma in erythrocyte media is also far from indifferent to the patient. Incompatibility of the donor and recipient with respect to plasma proteins is manifested by allergic, anaphylactoid and anaphylactic complications. The risk group in this regard consists of patients with IgA deficiency. In connection with the above, in order to reduce the reactogenicity and immunogenicity of erythrocyte media, they are subjected to additional “cleaning” by washing or leukofiltration in cases where they are used in transfusion therapy programs for hematological patients, in patients with an unfavorable transfusiological and/or allergic history, in children and obstetric practice.
However, the disadvantage of a suspension of washed erythrocytes, even with repeated washing, is the presence in it of residual donor leukocytes (at least 30%) in the absence of a practically significant admixture of plasma, and leukofiltered erythrocytes - the presence of plasma and, to a large extent, platelets. Only a combination of these two methods makes it possible to obtain an erythrocyte medium that is maximally purified from plasma proteins, antibodies, cytokines, platelets, and from leukocytes and their fragments.
The purpose of this study was to develop a method for obtaining and study the possibility of preserving an erythrocyte medium that is maximally depleted of other blood components. Taking into account the results of our previous studies on the creation of a leukofilter and on washing erythrocytes from plasma, as well as literature data, it seemed reasonable to remove leukocytes from the erythrocyte mass (EM) at the first stage of preparing a new medium, and at the second - washing the red cells. With this sequence of procedures, more favorable conditions are created for the retention of leukocytes during filtration (in the presence of plasma) and it becomes possible to exclude the removal of the leukothrombolayer with the underlying layer of red blood cells from the laundering technology, which reduces the loss of red cells
Materials and methods
To filter EM, we used the first domestic “Leukofilter device for removing leukocytes from preserved blood and erythrocyte media ULL-01” produced by NPP Interoko CJSC (TU 94444-031-17121966-99). For the preparation of leukofiltered washed red blood cells, the use of this device is more appropriate than the use of the new domestic leukosep filter.
In accordance with the “Device Operation Manual”, 50-100 ml of isotonic sodium chloride solution was injected into a container with EO obtained from canned donor blood for 1-2 days of storage (removal of the buffy coat is not required) and thoroughly mixed with erythrocyte medium. A washing liquid (isotonic sodium chloride solution) from a corresponding container in a volume of about 150 ml was passed through the filter assembly, trying to completely displace the air from the filter housing and washing it. Then the filter housing was filled with red blood cell medium, displacing the washing solution from it into a sterile container, which was then removed.
Leukofiltration was performed at a rate of 44.7±7.67 ml/min. The filtration procedure was completed by displacing the remaining filtered medium from the leucofilter body and filter material (passing 50 ml of isotonic sodium chloride solution) into a container with the filtered medium. The latter was then sealed and disconnected.
To wash leukofiltered erythrocytes, a special polymer container was used - “Container for washing erythrocytes by centrifugation” (TU 64-2-419-90, JSC Medical Preparations and Products “Sintez”, Kurgan). The container has a capacity of 500 ml, equipped with three polymer tubes with needles intended for connection to a container with erythrocyte medium to be washed, containers with a 0.9% sodium chloride washing solution and containers for draining the supernatant liquid after each centrifugation. Washing with centrifugation for 20 minutes at 2000 rpm (1250 g) with cooling was carried out twice. The supernatant was removed. Filtration and washing of erythrocytes were carried out under sterile conditions in a boxed room. Control for bacteriological contamination of suspensions was consistently negative.
To evaluate the effectiveness of the technology described above, we studied the content of leukocytes in the initial EM (in the Goryaev chamber), residual leukocytes (in the Nageotte chamber) and residual plasma protein in the resulting erythrocyte suspension (using Larionova’s reagent), as well as the loss of erythrocytes during the process of leukofiltration and washing. Washed erythrocytes without preliminary leukofiltration served as control.
Considering the high relevance of having reserves of the most “pure” erythrocyte medium for the blood service, in previous years we have developed a special erythropreservative Modelel-gluphosphate, which allows storing leukofiltered washed erythrocytes for 3 weeks. However, production of the drug Modelel was discontinued during the perestroika period and the organization of production of the erythropreservative Modelel-gluphosphate is currently impossible for objective reasons. In this regard, we have studied the fundamental possibility of storing leukofiltered washed red cells at 4°C using a modern preservative for native red blood cells SAGM (10 series of experiments). On the day of preparing the erythrocyte medium and then weekly, the morphofunctional usefulness of the erythrocyte suspensions was determined by examining the morphology of cells in the plasma (with the calculation of the morphological index), the content of free hemoglobin, the percentage of cell hemolysis, and the ATP content in them. The morphological index was calculated as the sum of the products of the percentage of individual forms of erythrocytes and the corresponding coefficients (100-200 cells were estimated). For discocytes, a factor of 1.0 was used; for mulberry forms - 0.6 and for spherocytes - 0.0 (modification of the Usry RT method). In addition, the percentage of osmotically unstable cells and the deformability of erythrocytes were studied (by the filtration method at constant pressure). The content of myeloperoxidase (MPO) and lactoferrin (LF) in the infusion of suspensions of washed erythrocytes was determined by ELISA at the Institute of Experimental Medicine of the Academy of Medical Sciences of the Russian Federation (Professor V.N. Kokryakov).
Results and discussion
The preliminary addition of an isotonic sodium chloride solution to the EM, its use for washing the filter material and for displacing residual media from the leucofilter after filtration, undoubtedly contribute to the effectiveness of further washing the filtered medium from plasma proteins. Rinsing the device with an isotonic sodium chloride solution before and after passing EM through it not only does not complicate the preparation of the proposed medium, but also reduces its losses (due to the leaching of red blood cells from the filter housing and connecting tubes).
The results of the study of the content of residual leukocytes and plasma proteins in the experimental and control series are presented in Table 1.
Table 1
Content of leukocytes and plasma proteins in erythrocyte suspensions
in the experimental (filtered washed) and control (washed)
series of experiments on the day of harvesting
| Series research | Content leukocytes in the original EM | Removal leukocytes, % | Content leukocytes in a dose of suspension | Content protein (g) in a dose of suspension |
| experience | 5.5 ± 1.09×109/l | 99,96 ± 0,005 | 0,55±0,065×106 | 0,06 ± 0,035 |
| control | 9.2 ± 1.21×109/l | 69,02 ± 6,023 | 0,86±0,170×109 | 0,05 ± 0,028 |
As can be seen from the data in table. 1, the proposed technology makes it possible to reduce the leukocyte content by 99.96±0.005% compared to the original erythromass. The residual number of white cells in a dose of a suspension of filtered washed erythrocytes immediately after its preparation averaged 0.55±0.065×106, which meets the requirements of international standards and provides reliable prevention of not only immunological non-hemolytic febrile reactions, but also alloimmunization, as well as the transmission of infections, caused by cytomegalovirus, Epstein-Barr virus, reduces the risk of transmission of the T-cell leukemia virus. In the control series, the content of leukocytes in the suspension dose turned out to be 3 orders of magnitude higher - on average 0.86 ± 0.170 × 109.
Double washing of erythrocytes sharply reduced the content of donor protein in the dose of suspensions both in the experiment (0.06±0.035 g) and in the control (0.05±0.028 g), which is an order of magnitude less than the value allowed by international standards (0.5 g/ dose). Therefore, filtered and washed red blood cells can be transfused into patients allergic to plasma proteins, IgA-deficient patients, and also those suffering from complement-dependent anemia.
The loss of red blood cells when using our proposed method was relatively small. They were equal in the experiment (when determined by a decrease in hemoglobin content) - 12.4 ± 0.20%; in the control, this figure turned out to be slightly lower (since there were no losses in the filtration unit) - 9.1±0.96%. However, even in the experimental series, the loss of red cells approached the level acceptable when preparing washed erythrocyte media (10-15%). Such losses are compensated by the advantages of the erythrocyte medium obtained by this method. The percentage of hemolysis as a result of the procedure changed slightly, the content of osmotically unstable erythrocytes decreased slightly. The ATP level corresponded to the initial level and was equal to 4.59±1.31 µmol/g Hb.
Early leukodepletion of erythrocyte media ensures, according to our data, the absence of an increase in the content of leukocyte breakdown products, in particular, myeloperoxidase and lactoferrin, during storage of erythrocyte suspension (Table 2).
In a series of filtered, washed erythrocytes in the erythropreservative Modelel-gluphosphate, the enzymes we studied were practically absent throughout the entire storage period. Only in one of the experiments, on the 21st day, extremely low levels of myeloperoxidase (23 ng/ml with a norm of 160±15 ng/ml) and lactoferrin (32 ng/ml with a norm of 1000±140 ng/ml) were found. At the same time, in suspensions of washed erythrocytes that were not subjected to leukofiltration, there was an increase in the concentration of MPO (from 42.7 ng/ml on the first day to 355.5 ng/ml by the end of the storage period) and LF (on average, from 580. 7 to 4242.5 ng/ml).
table 2
Content of MPO and LF in suspensions of filtered washed erythrocytes
(experiment) and washed erythrocytes (control) during storage at 4°C
| Indicators | Experience, days (n=4) | Control, day (n=4) |
| 1 14 21 | 1 14 21 | |
| Myeloperoxidase ng/ml (N = 160 ±15) | 0 ± 0,0 0 ± 0,0 5,8 ± 4,90 | 42,7 ±20,85 181,5 ± 94,32 355,5 ± 39,24 |
| Lactoferrin ng/ml (N = 1000 ±140) | 0 ± 0,0 0 ± 0,0 8,0 ± 6,90 | 580,7 ± 414,95 1412,5 ± 1054,60 4242,5 ± 714,20 |
Regarding the lactoferrin content, there was significant variability in the results between different experiments.
Thus, after two weeks of storage in the control series of experiments, its concentration ranged from 0 to 5046 ng/ml. The higher content of enzymes at the end of the storage period of unfiltered suspensions can be explained both by the washing technology in these series of experiments (without removing the buffy coat), which leads to an increased content of residual leukocytes in the suspension, and by the removal of plasma, which contains natural inhibitors of proteolytic enzymes.
To study the possibility of preserving leukofiltered washed erythrocytes at 4°C in a SAGM solution intended for native erythrocytes, 10 series of studies were carried out. Leukofiltration and washing of red cells were carried out in accordance with the “Temporary instructions for the method of preparing red blood cells, maximally devoid of leukocyte impurities (by filtration) and plasma (by washing)”, approved by the decision of the Academic Council of RosNIIGT.
Upon completion of leukofiltration and double washing of erythrocytes with isotonic sodium chloride solution, after removal of the supernatant, the erythropreservative SAGM (2:1) was added to the red cells of the experimental series. The resulting suspensions were stored at 4°C for 21 days. Indicators characterizing the morphofunctional properties of erythrocyte suspensions in the erythropreservative SAGM during their storage at 4°C are given in Table. 3.
The presented data indicate that by the end of the observed storage period of erythrocytes (21 days), the level of ATP remains quite high - more than 80% of the original - due to the content of adenine in the erythropreservative SAGM. However, during the storage of suspensions, a significant increase (more than 5 times by the 21st day) in the concentration of free hemoglobin and the percentage of hemolysis of red cells was noted. This does not allow us to recommend storing suspensions of filtered, washed erythrocytes for such a long time, despite the fact that the level of hemolysis has not reached the maximum permissible value (according to international standards - 0.8%), but to limit this period to one week, which is already important for activities blood service institutions. The organization in the future of the production of the erythropreservative Modelel-gluphosphate will provide the possibility of complete storage of leukofiltered washed erythrocytes for 21 days.
Table 3
Morphofunctional indicators of filtered suspensions
through leukofilter ULL-01 "Interoko" and washed erythrocytes
in erythropreservative SAGM (n=10) during storage at 4°C
| Shelf life, days | |
| Indicators | 1 7 14 21 |
| Morphological index | 97,5±0,98 88,0±4,72 87,0±2,83 84,4±5,90 |
| Hematocrit, % | 50,9±1,17 52,6±1,32 52,4±0,93 52,8±0,73 |
| Free hemoglobin, g/l | 0,30±0,038 0,72±0,167 1,36±0,219 1,72±0,222 |
| Hemolysis percentage | 0,09±0,009 0,21±0,043 0,41±0,060 0,51±0,071 |
| ATP content, µM/g Hb | 4,59±0,131 4,26±0,212 4,10±0,144 3,77±0,251 |
| ATP, percentage to the original | 100,0 92 ,8±3,99 89,5±3,79 82,2±4,91 |
| ONE, % | 0,9±0,58 1,6±0,43 4,2±0,83 7,7±2,59 |
| Deformability of erythrocytes IDE0 | 1,40± 0,508 1,21± 0,336 1,49± 0,378 1,80± 0,384 |
Conclusion
Filtration of erythrocytes through a domestic device-leukofilter ULL-01 produced by JSC NPP Interoko, followed by twice washing of filtered erythrocytes with isotonic sodium chloride solution, allows reducing the content of leukocytes to the level of 0.55±0.065×106 and protein to 0.06±0.035 g per dose. The medium prepared by the proposed method should be used within 24 hours, as provided by the current instructions for washed erythrocytes. Weighing in SAGM solution will extend its shelf life to 1 week. Taking this circumstance into account, it seems advisable to organize the production of polymer containers with SAGM solution, taking into account the justified needs for them.
A suspension of filtered, washed erythrocytes seems to be the optimal means of hemocomponent therapy for patients sensitized not only to plasma proteins, but also in the presence of anti-HLA, specific antigranulocyte and antiplatelet antibodies; transfusion-dependent patients to prevent alloimmunization and acute lung damage; immunocompromised persons to prevent the transmission of a number of viral infections; in pediatrics, including in the case of alloimmune thrombocytopenia of newborns; patients with a burdened obstetric and gynecological history (frequent pregnancies, miscarriages); for the prevention of “massive blood transfusions” and “allogeneic blood” syndromes
Interpretation
Reduced osmotic stability of red blood cells is characteristic of patients with hereditary spherocytosis. Spherocytes are osmotically less stable cells that are more easily lysed in a hypotonic solution compared to normal erythrocytes. The test for osmotic resistance after incubation of whole blood in a thermostat at t = 37 ° C is considered more sensitive than the test for osmotic resistance of red blood cells without incubation. Reduced osmotic resistance can also be observed in other diseases, including hereditary stomatocytosis, hereditary pyropoikilocytosis, and congenital dyserythropoietic anemia type II. This phenomenon can also be associated with autoimmune hemolytic anemia, poisoning, burns, and conditions after blood transfusion. Increased osmotic resistance of red blood cells indicates the presence of flattened red blood cells (leptocytes), which have a reduced surface area to volume ratio. This change occurs in iron deficiency anemia, thalassemia, and sickle cell disease because red blood cells with low hemoglobin content (MCH) and low mean cell volume (MCV) are unusually resistant to osmotic lysis. Reticulocytes and red blood cells from splenectomized patients are also more osmotically stable compared to normal cells. In liver diseases, due to passive accumulation of lipids, red blood cells are also more resistant to osmotic lysis.